The FreeStyle Libre portfolio is associated with improved clinical outcomes*1-9, reduced resource utilization*10-15, and cost savings†16.
Dr. Eden Miller explains how the FreeStyle Libre 3 system helps her to better manage diabetes in her practice.
Data-driven decision making
The FreeStyle Libre portfolio brings together an ecosystem of digital tools and features that members, providers, and caregivers can rely on.
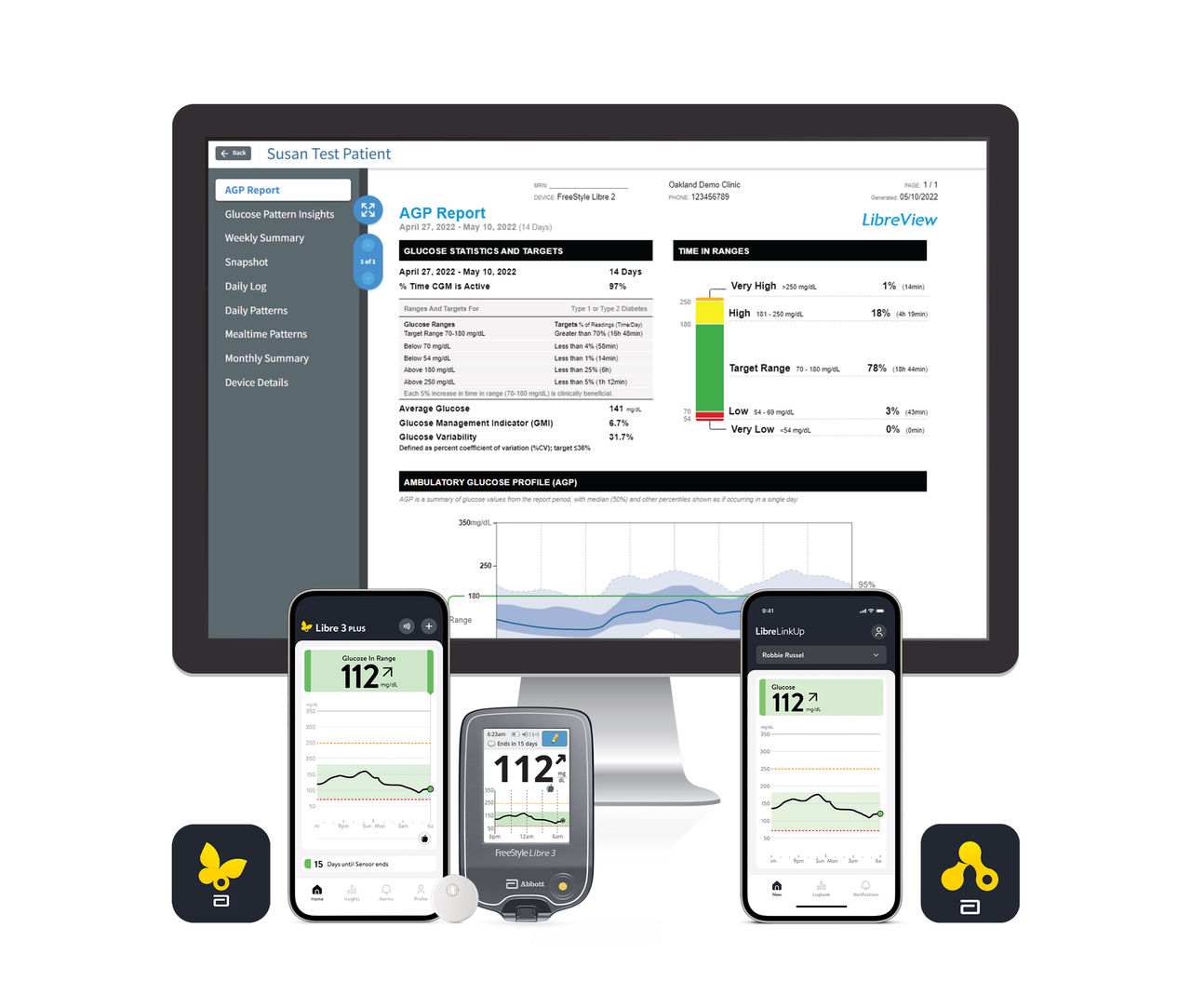
See how use of the FreeStyle Libre portfolio is associated with:
• Reduced HbA1c*1-7,18,19
• Reduced hypoglycemic events*8,9
• Reduced resource utilization*10-15
• Reduced diabetes-related work absenteeism*12,14

The little device that saves big†16.
In 2022, the total estimated cost of diagnosed diabetes was $412.9 billion17, with 1 in 4 healthcare dollars spent on people with diabetes. Since then, the use of the FreeStyle Libre portfolio has been associated with:
• Reduction in diabetes-related hospital admissions*14
• Reduction in hypoglycemic events*8,9

Continuous glucose monitoring (CGM) systems are small, wearable, sensor-based systems that help members identify glycemic patterns without fingersticks‡ so they can:
• Better manage diabetes*7-9
• Experience fewer complications*14
• Reduce diabetes-related work absenteeism*12,14

Visit our employer page to find reliable sources of health information and resources for your employees with diabetes.

Check out our FreeStyle Libre portfolio resources
Market Access Resources
HCP Resources
Member Resources
Stay up to date on the FreeStyle Libre portfolio.
Get the latest industry news, updates, and announcements, plus find out all there is to know about the FreeStyle Libre portfolio of products.
Product images are for illustrative purposes only. Not actual patient data.
Medicare and other payor criteria may apply. Abbott provides this information as a courtesy and does not guarantee payment or coverage.
The FreeStyle Libre 2 system and FreeStyle Libre 3 system are indicated for use in people with diabetes ages 4 and older.
FreeStyle Libre 3 Plus sensors are indicated for use in people with diabetes 2 and older.
The FreeStyle Libre systems apps are only compatible with certain mobile devices and operating systems. Please check the Support section of our website for more information about device compatibility before using the apps. Use of the FreeStyle Libre systems apps may require registration with LibreView.
The LibreLinkUp app is only compatible with certain mobile device and operating systems. Please check http://www.librelinkup.com for more information about device compatibility before using the app. Use of the LibreLinkUp app requires registration with LibreView. LibreLinkUp is not intended to be used for dosing decisions. The user should follow instructions on the continuous glucose monitoring system. LibreLinkUp is not intended to replace self-monitoring practices as advised by a physician.
The LibreView data management software is intended for use by both patients and healthcare professionals to assist people with diabetes and their healthcare professionals in the review, analysis and evaluation of historical glucose meter data to support effective diabetes management. The LibreView software is not intended to provide treatment decisions or to be used as a substitute for professional healthcare advice.
* Study was performed with the outside US version of the FreeStyle Libre 14 day system. Data is applicable to FreeStyle Libre 2 and FreeStyle Libre 3 system, as feature sets are similar as FreeStyle Libre 14 day system, excluding alarms.
† Based on prescription claims for commercial patients starting FreeStyle Libre Personal CGM systems compared to competitor CGMs. Does not include Medicare, Medicaid, and uninsured patients. The actual cost to patients may or may not be lower than other CGM systems, depending on the amount covered by insurance, if any.
‡ Fingersticks are required if your glucose alarms and readings do not match symptoms or when you see Check Blood Glucose symbol during the first twelve hours..
References: 1. Carlson AL, et al. BMJ Open Diabetes Res Care (2022): https://doi.org/10.1136/bmjdrc-2021-002590 2. Evans M, et al. Diabetes Ther (2022): http://doi.org/10.1007/s13300-022-01253-9 3. Wright, E. Diabetes Spectr (2021): https://doi.org/10.2337/ds20-0069 4. Kroger, J. Diabetes Care (2019): https://doi.org/10.1007/s13300-019-00741-9 5. Tyndall, V. Diabetologia (2019): https://doi.org/10.1007/s00125-019-4894-1 6. Yaron, M. Diabetes Care (2019): https://doi.org/10.2337/dc18-0166 7. Campbell, F. Pediatr Diabetes (2018): https://doi.org/10.1111/pedi.12735 8. Haak, T. Diabetes Ther (2017): https://doi.org/10.1007/s13300-016-0223-6 9. Bolinder, J. The Lancet (2016): https://doi.org/10.1016/s0140-6736(16)31535-5 10. Bergenstal, R. J Endocr Soc (2021): https://doi.org/10.1210/jendso/bvab013 11. Miller E, et al. AJMC (2021): https://doi.org/10.37765/ajmc.2021.88780 12. Charleer S, et al. Diabetes Care (2020): https://doi.org/10.2337/dc19-1610 13. Deshmukh, H. Diabetes Care (2020): https://doi.org/10.2337/dc20-0738 14. Fokkert, M. BMJ Open Diabetes Res Care (2019). https://doi.org/10.1136/bmjdrc-2019-000809 15. Kerr M, et al. Poster presented at the ATTD Conference, Madrid, Spain, February 19-22, 2020. http://doi.org/10.1089/dia.2020.2525.abstracts 16. Data on file, Abbott Diabetes Care. 17. American Diabetes Association. Diabetes Care (2023): https://doi.org/10.2337/dci23-0085
ADC-76905 v4.0
Important Safety Information
FreeStyle Libre 14 day, FreeStyle Libre 2 and FreeStyle Libre 3 systems: Failure to use FreeStyle Libre systems as instructed in labeling may result in missing a severe low or high glucose event and/or making a treatment decision, resulting in injury. If glucose reading and alarms (if enabled) do not match symptoms or expectations, use a fingerstick value from a blood glucose meter for treatment decisions. Seek medical attention when appropriate or contact Abbott at 855-632-8658 or FreeStyleLibre.us for safety info.
The sensor housing, FreeStyle, Libre, and related brand marks are marks of Abbott. Other trademarks are the property of their respective owners.
No use of any Abbott trademark, trade name, or trade dress in this site may be made without prior written authorization of Abbott Laboratories, except to identify the product or services of the company.
This website and the information contained herein is intended for use by residents of the United States.
ADC-85718 v5.0



